For years, medical devices have been manufactured using conventional technologies and processes such as molding, stamping and laser welding. However, these earlier manufacturing methods lacked customization and were limited to mass production. Advances in technology and new manufacturing techniques have significantly increased the potential for personalized medical devices, which directly impact patient outcomes.
3D printing, a form of additive manufacturing, creates objects by progressively building up thin layers of material. The main purpose of 3D printing is to turn digital designs into real-world objects. Typically, a 3D digital file from computer-aided design (CAD) or a magnetic resonance image (MRI) is employed to produce the 3D medical device. The US FDA has also provided detailed guidelines for medical device 3D printing and additive manufacturing.
The estimated market potential for biomedical devices is expected to reach $6.9 billion globally by 2028 (MarketsandMarkets™).
Benefits of 3D printing
This method of manufacturing brings substantial value to the global medical device industry:
Improved patient outcomes
3D printing enables the creation of patient-specific organ replicas, which offer higher adaptability after surgical or medical procedures and support faster recovery rates. The design and development of prosthetic devices allows for patient-specific implants and surgical guides.
Upgraded aesthetics
Whether producing surgical or prosthetic devices or implants, 3D printed devices can be customized for aesthetic suitability, improving patient lifestyle.
Decentralized production
3D printers provide flexibility in production volume and portability, unlike mass production processes that require specialized tooling. Devices can be produced in limited quantities without special tools and manufacturing can be brought to hospitals and remote locations.
Reduced cost of care and compliance
Mass production involves high equipment costs and less flexibility. 3D printing supports low-volume, specialized device manufacturing, facilitates paperless compliance and reduces human error.
Enhanced healthcare training
Training healthcare professionals with 2D images has limitations. 3D printed medical organs provide comprehensive training with greater scope for experimentation.
The next wave of Industry 4.0 technologies is reshaping 3D printing in MedTech — with GenAI emerging as a catalyst for innovation.
GenAI has the potential to revolutionize medical device 3D printing by executing thousands of design scenarios and virtual stress tests during production. It relies equally on software and hardware, playing a key role in overall product quality and outputs.

3D printing framework
The custom manufacturing process for medical devices begins with ingesting patient-specific data. The accuracy of manufacturing is directly tied to data precision. High-precision image scanners are typically used in the design phase of personalized medical devices.
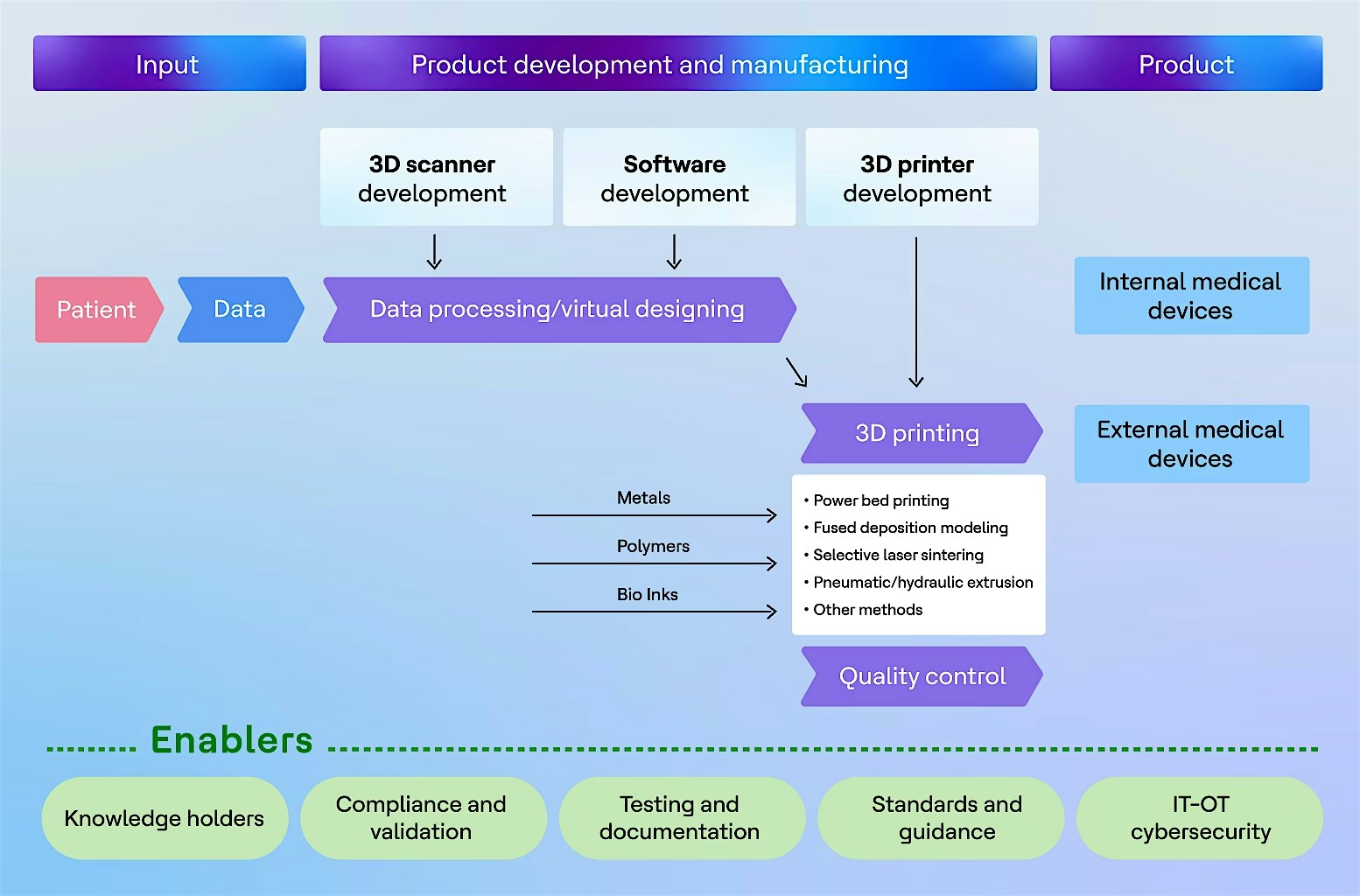
Before data is sent to the 3D printer, it is cleansed and precision designs are validated for anomalies. The printing technology and processes required for the device are determined in advance. Once data is ready, the 3D printer utilizes raw material to create the final product. The framework ensures digital compliance with strict adherence to US FDA Part 820, ISO 13485 and EU MDR guidelines for medical devices. Digital compliance enables error-free operations and robust quality standards. Knowledge management, cybersecurity, compliance, testing, best practices and standards are essential for deploying this modern manufacturing technology.
Challenges
3D printing brings its own set of challenges. Limited material selection (polymers, composites, metals and others) remains significant, as each 3D printer supports only certain raw materials, making material choice critical. For medical devices, the cost of 3D printers and equipment is substantial and must be accounted for in the final product pricing. Training the workforce to operate these complex systems also requires significant effort.
However, the industry is developing innovative composite materials such as specialty polymers and composites to accommodate diverse use cases.
Conclusion
The adoption of 3D manufactured medical devices is accelerating due to the ease of customization and improved patient outcomes. These factors are encouraging medical device organizations to address more use cases, gain competitive advantages and enhance profitability. This Industry 4.0 technology, combined with innovations in GenAI, has the potential to transform the sector and deliver next-generation medical devices.
References:
- Website article - nTop (Jan, 2023). What are 3-D printed medical devices. Retrieved on Oct 05, 2023 from https://www.ntop.com/resources/blog/what-are-3d-printed-medical-devices/
- Website US FDA (May, 2023). 3D Printing of Medical Devices. Retrieved on Oct 13, 2023 from https://www.fda.gov/medical-devices/products-and-medical-procedures/3d-printing-medical-devices
- Generative AI – Kishor K (May, 2017). Generative AI is Revolutionizing 3D Printing. Retrieved on Oct 23, 2023 from https://medium.com/@Nontechpreneur/generative-ai-is-revolutionizing-3d-printing-8605c3c080ff